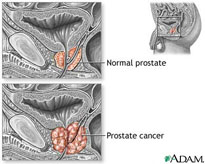
Prostate Cancer
Prostate cancer is the most commonly diagnosed cancer in the Western world. The five-year worldwide prevalence is estimated to be in excess of 1.5 million1 with an annual incidence of approximately 670,000.2 The incidence tends to be highest in Western countries and the lowest in Asia: 3
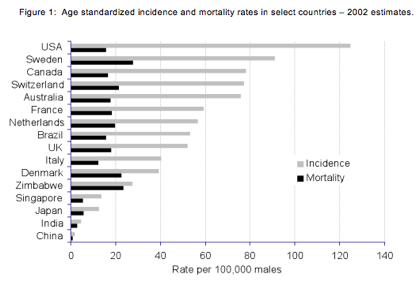
The geographic variability in incidence is thought to be due to a number of factors including genetics, diet, and detection, but despite the lower overall incidence in Asian countries, incidence has risen sharply in some Asian countries recently.4
Virtually all prostate cancer is initially hormone-sensitive, meaning growth is promoted in the presence of androgens like testosterone and its more potent metabolite dihydrotestosterone.5 Accordingly, the most common pharmacological treatment for the cancer is so-called “androgen-deprivation therapy” or ADT, which aims to lower a patient’s testosterone levels to levels equivalent to that obtained by surgical castration, also know as orchiectomy.6
ADT can be accomplished in a number of ways and the original non-surgical therapy was implemented using estrogens, specifically the very potent synthetic estrogen diethylstilbestrol or DES.7 DES works by inhibiting the release, through a feedback mechanism, of gonadotropin-releasing hormone (GnRH, also known as luteinizing hormone releasing hormone or LHRH) in the hypothalamic area of the brain.8 The ensuing inhibition of gonadotropin release blocks the gonadal production of testosterone and its metabolites, particularly dihydrotestosterone (DHT).9
For a number of years DES was the standard of care for hormone-sensitive disease, but it fell from favor when the therapy was associated with a significant level of thromboembolic and other cardiovascular events.10 With the discovery of analogs of GnRH that could block gonadal steroid hormone production in a manner analogous to DES, this approach began to gain favor, and today leuprolide and other GnRH agonists are the standard of care for hormone-sensitive disease.11
GnRH agonists are not without their problems, however. One issue relates to the fact that they are, in fact, agonists and not antagonists of GnRH. This means that they are designed to “turn on” gonadotropin and steroid secretion, rather than to turn it off. When given continually, rather than in a pulsatile fashion, they increase gonadotropin and sex steroid output, but later they inhibit the gonadotropin/steroid secretion by de-sensitizing the GnRH receptor in the pituitary.12 So, while the net effect is suppression of GnRH and sex hormone production, there is an initial surge of testosterone when treatment starts and this can cause tumor flare.13 In order to avoid this flare, it is common for physicians to co-prescribe a second class of agents called “anti-androgens” that block testosterone activity at the androgen receptor level.14 Thus, despite the fact that the patient gets high transient levels of testosterone produced through the action of the GnRH agonist, androgens are blocked from acting by the presence of this adjunct agent.15
It is increasingly recognized that the GnRH agonists are associated with other substantial morbidities, perhaps the most serious of which is osteoporosis.16 Because these agents block all sex hormone production, including estrogens, and estrogens are principally responsible for bone maintenance, these estrogen-deprived patients lose bone at an alarming rate – from 4% to 13% per year of GnRH agonist therapy – and this results in an exponential increase in fracture risk.17
Patients on GnRH agonist therapy also suffer from morbidities associated with obesity, altered lipid profiles, increased cardiovascular disease and cardiovascular death, and symptoms of metabolic disorder like increased insulin resistance.18
Reduced estrogen input to key brain areas also causes an increase in the number and frequency of vasomotor symptoms (“hot flashes”) with the associated discomfort and deteriorating quality of life.19 A hot flash is a sudden feeling of heat that can last for several minutes, followed by profuse sweating and occasional shivering. Sudden reddening of the skin on the head, neck, and chest can occur due to increases in blood flow, conductance, and temperature. Palpitations, sleep disturbances, skin blotching, and anxiety can also accompany hot flashes.20,21 In men receiving ADT, these hot flashes tend to continue throughout therapy22. As with postmenopausal women, estrogens are thought to act at the hypothalamic thermoregulatory and vasomotor centers, and thus are used therapeutically to eliminate the incidence and magnitude of hot flashes that occur with the loss of gonadal steroids in men undergoing ADT with GnRH analogs.23
Our approach to prostate cancer is several-fold. First, we intend to develop our proprietary transdermal estradiol formulation in the primary treatment of hormone-sensitive prostate cancer, competing directly with the GnRH agonists. Data suggest that by delivering estradiol transdermally, rather than orally, the thromboembolic events seen with earlier estrogen therapies will be substantially mitigated; moreover, using estrogen to suppress GnRH activity and androgen output should avoid the bone loss and thermoregulatory problems associated with the GnRH agonists.24
Second, we intend to develop our proprietary formulation for adjunct therapy, co-administered with the current GnRH products. We believe that this adjunct therapy will take advantage of the bone-sparing effects and control of vasomotor symptoms offered by the estradiol, while relying on the androgen-deprivation effects of the GnRH products. I.e., for those patients who have begun GnRH therapy, BHR-200 has the potential to offer relief from the hot flashes caused by estrogen loss and the diminished quality of life due to these episodes by replacing the estrogen lost during treatment.
Finally, we have in our pipeline a selective estrogen receptor modulator and we intend to utilize its antiestrogenic properties to prevent and treat gynecomastia – the benign enlargement of the male breast, usually associated with an estrogen/androgen imbalance – which is a side effect of androgen deprivation therapy.
1 http://www.prostateline.com/prostatelinehcp/9898_12031_4_1_0.aspx
2 UK Prostate Cancer incidence statistics; Prostate Cancer UK.
4 http://www.prostateline.com/prostatelinehcp/9898_12031_4_1_0.aspx
5 Hormone (Androgen Deprivation) Therapy; American Cancer Society.
7 Crawford, Rev Urol. 6 (suppl 7):S3-S11 (2004).
16 Guise et al., Rev Urol. 9:163-180 (2007).
20 Spetz et al., J Support Oncol. 1 :263-273 (2003).
21 Alekshun and Patterson, Supportive Cancer Therapy 4(1):30-37 (2006).
22 Guise et al., Rev. Urol. 9(4) :163-180 (2007).
23 Higano, Urology. 61(2 suppl 1):32-38 (2003).
24 Ockrim et al., Nature Clinical Practice Oncology 3:552-63 (2006).